Cucumis melo (PROTA)
Introduction |
| General importance | |
| Geographic coverage Africa | |
| Geographic coverage World | |
| Cereal / pulse | |
| Vegetable | |
| Vegetable oil | |
| Medicinal | |
| Ornamental | |
| Forage / feed | |
| Food security | |
Cucumis melo L.
- Protologue: Sp. pl. 2: 1011 (1753).
- Family: Cucurbitaceae
- Chromosome number: 2n = 24
Vernacular names
- Melon, muskmelon, cantaloupe (En).
- Melon (Fr).
- Melão (Po).
- Mtango, mtango mungunyana, mmumunye (Sw).
Origin and geographic distribution
Melon probably originated in East Africa, where wild populations still occur, e.g. in Sudan, Ethiopia, Eritrea, Somalia, Uganda and Tanzania. Possibly it also occurs wild in southern Africa, but the exact distribution of wild Cucumis melo is unclear because of the regular occurrence of plants escaped from cultivation. Melon was domesticated in the eastern Mediterranean region and West Asia at least 4000 years ago and subsequently spread into Asia. During the long period of cultivation many types developed with many fruit shapes and with either sweet or non-sweet flesh. Important centres of genetic diversity of cultivated melon developed in Iran, Uzbekistan, Afghanistan, China and India. In Africa important variation occurs in Sudan and Egypt. The name ‘cantaloupe’ derives from a 15th century introduction of melon from Turkish Armenia to the papal residence at Cantalupi near Rome. Melon is now grown worldwide. It is a typical fruit vegetable of subtropical and warm temperate areas.
Melon occurs throughout the warm and dry areas of Africa, where it is grown either for its fruit or for its seeds.
Several non-sweet types of Cucumis melo are grown traditionally. The most important one is snake melon, called ‘ajjur’, ‘faqqus’ or ‘qatta’ in Arabic. It is found in many parts of Asia, from Turkey to Japan, and locally in Europe (Italy) and the United States. In Africa it seems to be restricted to Sudan and North Africa (Egypt, Morocco, Tunisia), where it is quite important. In Sudan the immature fruits of a melon type locally known as ‘tibish’ are used in the same way as snake melon. Some other types grown in Africa have bitter flesh, and are grown for their edible seeds.
Uses
Mature fruits of sweet melon cultivars are usually consumed fresh for the sweet and juicy pulp. The pulp is also mixed with water and sugar, or sometimes with milk, and served as a refreshing drink or made into ice cream. Immature fruits of non-sweet types, including snake melon, are used as a fresh, cooked or pickled vegetable; they are also stuffed with meat, rice and spices, and fried in oil. Snake melon is often confused with cucumber and used as such. The seeds are eaten after roasting; they contain edible oil. The Hausa people in Nigeria grind the kernels to a paste and make it into fermented cakes. The young leaves are occasionally consumed as a potherb and in soups. The leafy stems and also the fruit provide good forage for all livestock. In Réunion and Mauritius a decoction of seeds and roots is used as a diuretic and vermifuge.
Production and international trade
Annual world production of melon has increased from 9 million t (700,000 ha) in 1992 to 22 million t (1.2 million ha) in 2002. Major producing countries are China with 400,000 ha, West Asia (Turkey, Iran, Iraq) 200,000 ha, the Americas (United States, Mexico, Central and South American countries) 165,000 ha, northern Africa (Egypt, Morocco, Tunisia) 110,000 ha, southern Asia (India, Pakistan, Bangladesh) 100,000 ha, European Union (Spain, Italy, France, Greece, Portugal) 95,000 ha, Romania 50,000 ha, Japan 13,000 ha and Korea 11,000 ha.
Each country has its own specific melon cultivars and most of the crop is sold in local markets. Production for export has developed in the Mediterranean region, the United States, Mexico, Australia, Taiwan and Japan, using F1 hybrid cultivars with good shipping and storage characteristics.
In Africa sweet melon is a luxury crop for urban markets, grown in drier regions and in highlands. Statistics on production are not available for most countries, except Cameroon (3500 ha) and Sudan (1200 ha). Senegal and surrounding countries export melon during the winter to Europe.
Snake melon is important in Sudan, where it is grown for home use and local markets. The area grown is about 4000 ha with an annual production of 80,000 t. It is not exported.
Properties
The edible portion of a mature melon fruit is 45–80%. Fruits (raw, peeled) contain per 100 g edible portion: water 90.2 g, energy 142 kJ (34 kcal), protein 0.8 g, fat 0.2 g, carbohydrate 8.2 g, fibre 0.9 g, Ca 9 mg, Mg 12 mg, P 15 mg, Fe 0.2 mg, Zn 0.2 mg, vitamin A 3382 IU, thiamin 0.04 mg, riboflavin 0.02 mg, niacin 0.7 mg, folate 21 μg, ascorbic acid 37 mg (USDA, 2002).
The nutritional composition of snake melon per 100 g edible portion is: water 94.5 g, energy 75 kJ (18 kcal), protein 0.6 g, fat 0.1 g, carbohydrate 4.4 g, fibre 0.3 g, ascorbic acid 13 mg (Polacchi, W., MacHargue, J.S. & Perloff, B.P., 1982).
Sugar content and aroma are important factors determining the quality of sweet melon. Esters derived from amino acids are important components of the characteristic flavour; sulphur-containing compounds also play a role. Several C-9 alcohols and aldehydes, including Z-non-6-enal, are characteristic of the melon aroma. To get the best aroma fruits should be harvested only 2–3 days before they are fully ripe. The edible seed kernel contains approximately 46% of a yellow oil and 36% protein.
Adulterations and substitutes
Snake melon for use in salads can be replaced by cucumber (Cucumis sativus L.). As a fruit, sweet melon can be replaced by papaya.
Description
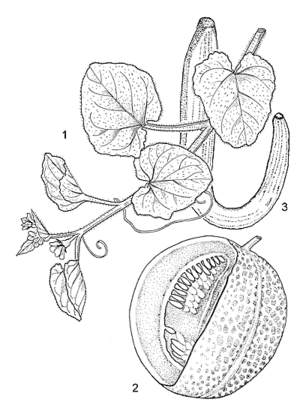
- Monoecious, climbing, creeping or trailing, annual herb, having simple tendrils; root system large, mostly distributed in the top 30–40 cm of the soil, a few roots descending to 1 m depth; stem up to 3 m long, ridged or striate, hairy.
- Leaves alternate, simple; stipules absent; petiole 4–10 cm long; blade orbicular or ovate to reniform, 3–15(–20) cm in diameter, angular or shallowly palmately 5–7-lobed, cordate at base, shallowly sinuate-toothed, surfaces hairy.
- Flowers axillary, unisexual or bisexual, regular, 5-merous; pedicel 0.5–3 cm long; sepals linear, 6–8 mm long; corolla campanulate, lobes almost orbicular, up to 2 cm long, yellow; male flowers in 2–4-flowered fascicles, with 3 free stamens; female or bisexual flowers solitary, with inferior, ellipsoid ovary, stigma 3-lobed.
- Fruit a globose, ovoid or oblongoid berry weighing 0.4–2.2 kg, smooth or furrowed, rind smooth to rough and reticulate, white, green, yellowish-green, yellow, yellowish-brown, speckled yellow or orange with green or yellow background, flesh yellow, pink, orange, green or white, many-seeded.
- Seeds compressed ellipsoid, 5–12 mm × 2–7 mm × 1–1.5 mm, whitish or buff, smooth.
- Seedling with epigeal germination.
Other botanical information
Most of the about 30 Cucumis species are native to Africa. They all have a chromosome number of 2n = 24, except Cucumis sativus L. (cucumber) with 2n = 14; probably this species originated from Asia. Cucumis sativus fruits are beset with spinous tubercles and warts when young, whereas ovaries of Cucumis melo are hairy without tubercles and warts.
Cucumis melo is polymorphic. Wild and weedy plants are often distinguished as subsp. agrestis (Naudin) Pangalo, having shortly pubescent ovaries and comparatively small flowers and fruits, whereas the cultivated plants (subsp. melo) have villous ovaries and generally larger flowers and fruits.
Cultivated plants belong to many different cultivar-groups, of which the most important with sweet fruits for modern market gardening are:
– Reticulatus Group (muskmelon or netted melon): fruit globular (1–1.8 kg), rind strongly reticulate, sometimes furrowed, yellowish-green with orange flesh (Italo-American) or finely reticulate to smooth, yellowish-green with pale green flesh (Japanese, Mediterranean, e.g. ‘Galia’), sugar content high (13–15%), aromatic, shelf life medium.
– Cantaloupe Group (cantaloupe or muskmelon): fruit flattish to globular and often ribbed (1.2–1.8 kg), rind smooth or reticulate, flesh usually orange, carotene and sugar content high, flavour rich, shelf life short, mainly grown in south-western Europe (e.g. ‘Charentais’) and the Americas.
– Inodorus Group (winter melon): fruit ovoid (1.5–2.5 kg), late maturing, rind smooth, wrinkled or slightly reticulate, often striped or splashed, grey, green or yellow, flesh firm, white or pale green, sugar content high but little flavour, shelf life long, mainly grown in Iran, Afghanistan and China, but also in Spain, the United States and Japan; important cultivars are ‘Casaba’, ‘Honeydew’, ‘Piel de Sapo’, ‘Jaune Canari’ and ‘Chinese Hami’.
Examples of groups with non-sweet fruits used as a vegetable in Africa are:
– Flexuosus Group (snake melon or ‘snake cucumber’): fruit up to 2 m long, more than 6 times as long as wide, rind pale green or striped pale and dark green, ribbed or wrinkled, flesh white.
– Tibish Group: fruit small, ovoid to oblate, without ribs, rind smooth, dark green with pale green stripes, flesh firm, white, particularly important in Sudan; a similar type named ‘seinat’ in Sudan is grown for its seed.
Sweet melon grown for the urban markets in tropical Africa comprises nowadays mostly F1 hybrid cultivars of Reticulatus Group (e.g. ‘Galia’) and Cantaloupe Group (e.g. ‘Charentais’).
Growth and development
Melon seed will remain viable for at least 6 years when stored dry (moisture content 6%) at temperatures below 18°C. Priming may improve germination after long storage. Seedlings appear (2–)4–8(–14) days after sowing. Numerous horizontal lateral roots develop rapidly from the taproot. The roots grow mostly at a depth of 30–40 cm. The first true leaf appears 5–6 days after unfolding of the cotyledons. The first 2–4 axillary buds on the main stem produce vigorous primary branches, which check the growth of the main stem. In most types, the first clusters of male flowers appear on the 5th–12th node of primary branches, while bisexual or female flowers appear on tertiary branches, formed from the 14th node of primary branches onwards. Flowers are open for one day only and insects, mostly bees, effect pollination. The fruit is a heavy sink for assimilates and minerals and per plant usually only 3–6 fruits will develop out of 30–100 female/bisexual flowers. The fruit development curve is sigmoid with maximum growth at 10–40 days after flowering; maturation with little further expansion occurs during the last 10 days when sugars accumulate in the fruit flesh and the net tissue on the fruit surface develops. Fruits mature 75 days (early cultivars of Reticulatus Group and Cantaloupe Group) to 120 days (Inodorus Group) after sowing. In the fruits of Reticulatus Group and Cantaloupe Group ethylene plays an essential role in the ripening process (climacteric), e.g. for flesh softening, yellowing of the rind and abscission from the pedicel. Shelf life of these fruits is short (< 1 week for ‘Charentais’) to medium (2–3 weeks for ‘Galia’). Fruits of cultivars of Inodorus Group do not produce ethylene during ripening (non-climacteric) and consequently have a long 3 months for ‘Piel de Sapo’). Ethylene-independent ripening processes are flesh coloration, and accumulation of sugars and organic acids.
Ecology
Wild Cucumis melo plants occur in open woodland, especially along rivers, and as a weed in fields and waste places, up to 1200 m altitude.
Melon requires warm and dry weather with plenty of sunshine for growth and production. The optimum temperature range is 18–28°C, growth being severely retarded below 12°C. Melon easily withstands several hours per day of very high temperatures, up to 40°C. Plants are killed instantly by frost. In snake melon stem elongation was found to be greater under short 8-hour days than under 16-hour days. High humidity will reduce growth, adversely affect fruit quality and encourage leaf diseases. Melon grows best on deep, well-drained and thoroughly cultivated fertile loamy soils with pH 6–7. It does not tolerate very acid soils or waterlogged soil.
Propagation and planting
Melon is usually direct-seeded: 2–3 seeds per hole, sown 2–4 cm deep on mounds or ridges, later thinned to one plant. Spacing is 50–60 cm within and 120–200 cm between the rows, giving a density of 8000–16,000 plants per ha. Alternatively, seedlings are raised in polythene pots or in soil blocks and transplanted carefully to the field when 4 weeks old, taking care not to damage the root system. The weight of 1000 seeds is (8–)25–35 g. Seed rates per ha are 1.5–2 kg for direct-seeded melon and 0.5 kg for the transplant method. In Sudan snake melon is sown directly on raised flats of 2 m wide, in holes on both sides of the flats.
Management
Melon can be grown in normal upland conditions, provided that it is rotated with non-cucurbit crops to avoid soilborne diseases and nematodes. The soil should be ploughed, harrowed and rotavated to attain a well-pulverized and well-levelled soil. Furrow or drip irrigation is common, since plants have a high demand for water until the fruits have reached maturity. Melon is less dependent on daily irrigation than cucumber or pumpkin. During the dry season, one litre water per planting hole may be given. In Sudan an established snake melon crop is irrigated every 10–12 days during the hot rainy season (March–October), and every 14–18 days during the cool season.
Fertilizer requirements depend on crop performance and nutrient status of the soil. Removal of nutrients in a harvest of 20 t/ha of fruits is: N 60–120 kg, P 9–18 kg, K 100–120 kg, Ca 70–100 kg and Mg 10–30 kg. Melon responds well to organic manures applied at 25–30 t/ha. A complete fertilizer should be applied before sowing or planting, followed by a N topdressing when the stems are 20–30 cm long. Melon is particularly sensitive to Ca deficiency, which causes glassiness or watercore in the fruits. It is also sensitive to molybdenum deficiency, occurring in ferralitic soils. Melon is very sensitive to a number of herbicides, including Atrazine, and may even be damaged by herbicide residues from preceeding crops.
Mulching is a well-established practice in the production of melon. In subtropical areas, black, transparent or silver-painted polythene sheets are commonly used not only to control weeds but also to raise or lower the temperature of the soil. In the tropics, common mulching materials are rice straw or grass, and increasingly plastic foil. In areas where mulching materials are not available, weeding is necessary until the plants start producing long stems. Hand hoeing or pulling of large weeds is often practised. Various methods of pruning primary and secondary branches are applied to regulate vegetative growth and fruit set (3–5 fruits per plant). Snake melon is sometimes grown along a trellis to get straight fruits.
Diseases and pests
Several diseases may affect melon. Gummy stem blight (Didymella bryoniae) causes stem and fruit stalk canker, fruit rot and plant wilting. Is a serious disease in humid and hot conditions. It is controlled by the use of disease-free seed, seed disinfection, crop rotation, spraying fungicides, and especially planting resistant cultivars. Powdery mildew (Sphaerotheca fuliginea and Erysiphe cichoracearum) can be controlled by fungicides, but modern F1 hybrids have high tolerance to most types. Downy mildew (Pseudoperonospora cubensis) is important in hot and humid climates and can be controlled by fungicides; polygenically controlled resistance is available in certain Indian accessions. Anthracnose (Colletotrichum lagenarium) can be controlled by seed treatment, crop rotation and fungicides. Damping-off (Pythium sp. and Rhizoctonia sp.) has to be prevented by treating seed with fungicides. Bacterial soft rot or bacterial wilt (Erwinia tracheiphila) is controlled by removing affected plants and by eliminating the vector (striped and spotted cucumber beetle) with insecticide sprays. Angular leaf-spot (Pseudomonas lachrymans) is primarily a cucumber disease but occurs occasionally also in melon. Fusarium wilt (Fusarium oxysporum f.sp. melonis) can be effectively prevented only by resistant cultivars. In snake melon it is also a serious problem. It is most aggressive at lower temperatures (18–20°C) and does not occur in the lowlands. Sudden wilt caused by the soilborne fungus Monosporascus cannonballus has become a serious problem in areas with a subtropical climate, neutral or alkaline soils, high (30–35°C) soil temperatures and use of plastic mulch; it has not yet been recorded in tropical Africa but is a potential menace.
The most frequent virus in tropical conditions is the aphid transmitted papaya ringspot virus (PRSV-W, formerly WMV-1) for which good resistance is available. Cucumber mosaic virus (CMV), watermelon mosaic virus (WMV-2) and zucchini yellow mosaic virus (ZYMV), all three transmitted by aphids, in particular Aphis gossypii, affect melon and predominate in subtropical conditions; there are various sources of resistance to these three viruses and also to the vector. Other virus diseases in melon are muskmelon necrotic spot virus (MNSV), transmitted by the soil fungus Olpidium sp., the soil- and seedborne cucumber green mottle mosaic virus (CGMMV) and beet curly top virus (BCTV) transmitted by leafhoppers.
Root-knot nematodes (Meloidogyne spp.) can be a serious problem when melons are grown without proper crop rotation; control can be done by soil solarization or by wide-spectrum soil fumigants, but the latter are expensive and hazardous to the environment.
Pests in melon are thrips (Thrips palmi and Frankliniella spp.), spider mite (Tetranychus urticae), aphids (Aphis gossypii), melon fruit fly (Dacus cucurbitae), cucumber beetles (Diabrotica spp.), leaf folder (Diaphania indica), leaf feeder (Aulacophora similis) and the fly Bactrocera cucurbitae, which is especially active in the humid tropics and causes young fruits to drop by tunnelling in the pedicel. Farmers usually control these pests with insecticides. However, indiscriminate use of insecticides only aggravates pest problems by destroying useful parasitic insects.
Harvesting
Cantaloupe and muskmelon tend to separate from the pedicel at the base of the fruit at maturity due to the formation of an abscission layer. This is called ‘full slip’. Harvesting occurs usually at the ‘half slip’ stage. Fruits of Inodorus Group do not form an abscission layer and maturity is indicated by colour change, e.g. from green to yellow. Tender immature pale green fruits of snake melon are harvested starting 45–60 days from sowing. Harvested fruits are about 20 cm long with a diameter of 3 cm, weighing 90–100 g. If the fruits are left for seed, they are harvested when fully ripe.
Yield
On average yields of fresh melon fruits reach 18 t/ha, ranging from 5–40 t/ha depending on cultivar and cultural practices. Seed yields are about 300–500 kg/ha for open-pollinated and 100–200 kg/ha for hybrid cultivars. The yield of a snake melon crop in Sudan is on average 20 t/ha.
Handling after harvest
Muskmelon fruits for storage should be cooled to 10–15°C immediately after harvesting to retard ripening. Storage for 10–15 days at 3–4°C (90% relative humidity) is possible, but lower temperatures can cause chilling injury. ‘Honeydew’ and other winter melon fruits can be stored at 10–15°C for longer periods, some cultivars up to 90 days. Heavily netted melon fruits (e.g. the Mediterranean ‘Galia’ and American ‘Western Shipper’) are relatively resistant to handling and transport. The fruits of snake melon are packed in plastic or jute sacs for transport to near-by town markets. They are treated in the same way as cucumber fruits.
Genetic resources
The genetic diversity within Cucumis melo is fairly well preserved in germplasm collections. The most extensive collections are maintained in the United States (North Central Regional Plant Introduction Station, Ames IA), Russian Federation (N.I. Vavilov All-Russian Scientific Research Institute of Plant Industry, St. Petersburg), China (Institute of Crop Germplasm Resources (CAAS), Beijing), but many other countries hold significant collections. In Africa important collections are held at National Horticultural Research Institute, Ibadan, Nigeria and Agricultural Research Corporation, Wad Medani, Sudan. The melon germplasm collections preserved in Wad Medani include about 70 accesssions of snake melon and 45 of the local vegetable melon ‘tibish’. The germplasm collections could be complemented by further collection of germplasm in the secondary centres of genetic diversity in Afghanistan, India, China, Pakistan and Sudan. Some cultivar-groups with small fruits close to wild types appear to be particularly good sources of host resistance to major melon diseases.
Breeding
Much of the cultivar improvement in melon is based on mass and line selection in open-pollinated populations. However, these are now rapidly giving way to F1 hybrid cultivars, especially in Europe, the United States, Japan and Taiwan. Pure-line development in melon is easy, as there is practically no inbreeding depression after repeated selfing. On the other hand, there is also little hybrid vigour in hybrids between inbred lines. The main advantages of F1 hybrids are, however, uniformity of plant and fruit type and recombination of favourable characteristics of different melon types in one genotype: fruit quality (round shape, good flavour, high sugar content, small seed cavity), long shelf life, adaptation to more humid climates and especially resistance to diseases and pests.
Most cultivars have male as well as bisexual flowers on the same plant, and F1 hybrid seed production requires emasculation of the bisexual flowers followed by hand pollination. Monoecious plant types would enable hybrid seed production with bee pollination, as the female parent line can be temporarily induced to produce female flowers only by sprays with ethrel. However, the change to monoecious F1 hybrids is slowed down by the fact that monoecy in melon is linked to elongated shape and large size of the fruits, while the aim for cantaloupe and muskmelon is round and compact fruits. However, monoecious F1 hybrids are now becoming increasingly common.
The main breeding objectives for snake melon are disease and insect resistance and better fruit quality. Breeding for resistance is a top priority because the fruit is often consumed fresh, making it hazardous to use chemicals, especially where restrictions are not observed. Breeding for quality aims at producing cultivars with slender and tender fruits for the fresh consumption in green salads.
In a recent evaluation study of melon germplasm collected from Sudan, several snake melon accessions were found, including resistant plants to some types of Fusarium wilt and Sphaerotheca fuliginea. Crosses with normal melon are easy and occur spontaneously. Resistance to Fusarium wilt was also detected in some accessions of Tibish Group and wild populations of Cucumis melo, known locally in Sudan as ‘humaid’. Resistance to some viral diseases, especially ZYMV, was also detected in some accessions of ‘humaid’.
Prospects
Melon is well liked by most people and the importance of this crop will further increase through better adaptation to hot and humid growing conditions. A factor limiting melon production is the multitude of diseases (viruses in particular) and pests. New techniques from cellular (protoplast fusion) and molecular (genetic transformation, DNA markers) biology are now within reach of the melon breeders. This has opened up prospects for exploiting germplasm from other Cucumis species for disease and pest resistance and other characteristics, not available through conventional interspecific hybridization.
Snake melon is adapted to hot and dry conditions, which makes it interesting for further expansion to some African regions outside Sudan. Germplasm collection and breeding need more attention.
Major references
- Ezura, H., Akashi, Y., Kato, K. & Kuzuya, M., 2002. Genetic characterization of long shelf-life in Honeydew (Cucumis melo var. inodorus) melon. Acta Horticulturae 588: 369–372.
- Montforte, A.J., Garcia-Mas, J. & Arús, P., 2003. Genetic variability in melon based on microsatellite variation. Plant Breeding 122: 153–157.
- Odet, J. (Editor), 1991. Le melon. 2nd Edition. Centre Technique Interprofessionnel des Fruits et Legumes (CTIFL), Paris, France. 293 pp.
- Paje, M.M. & van der Vossen, H.A.M., 1993. Cucumis melo L. In: Siemonsma, J.S. & Kasem Piluek (Editors). Plant Resources of South-East Asia No 8. Vegetables. Pudoc Scientific Publishers, Wageningen, Netherlands. pp. 153–157.
- Pitrat, M. & Risser, G., 1992. Le melon. In: Gallais, A. & Bannerot, H. (Editors). Amélioration des espèces végétales cultivées. Institut National de la Recherche Agronomique (INRA), Paris, France. pp. 448–459.
- Pitrat, M., Hanelt, P. & Hammer, K., 2000. Some comments on infraspecific classification of cultivars of melon. Acta Horticulturae 510: 29–36.
- Robinson, R.W. & Decker-Walters, D.S., 1997. Cucurbits. CAB International, Wallingford, United Kingdom. 226 pp.
- Rubatzky, V.E. & Yamaguchi, M., 1997. World vegetables: principles, production and nutritive values. 2nd Edition. Chapman & Hall, New York, United States. 843 pp.
- Shinohara, S. (Editor), 1984. Vegetable seed production technology of Japan. Volume 1. Shinohara's Authorized Agricultural Consulting Engineer Office, Tokyo, Japan. 432 pp.
- Whitaker, T.W. & Davis, G.N., 1962. Cucurbits - botany, cultivation and utilization. Leonard Hill, London, United Kingdom. 249 pp.
Other references
- Andres, T.C., 2003. Web site for the plant family Cucurbitaceae and home of the cucurbit network. [Internet] http://www.cucurbit.org/family.html. May 2004.
- El Tahir, I.M. & Pitrat, M., 1999. Tibish, a melon type from Sudan. Cucurbit Genetics Cooperative Report 22: 21–23.
- Fisher, C. & Scott, T.R., 1997. Food flavours: biology and chemistry. The Royal Society of Chemistry, Turpin Distribution Services, Letchworth, United Kingdom. 165 pp.
- Jones, B., Pech, J.C., Bouzayen, M., Lelièvre, J.M., Guis, M., Romajaro, F. & Latché, A., 2001. Ethylene and developmentally-regulated processes in ripening climacteric fruit. Acta Horticulturae 553: 133–138.
- Karchi, Z., 2000. Development of melon culture and breeding in Israel. Acta Horticulturae 510: 13–17.
- Mirghani, K.A. & El Tahir, I.M., 1997. Indigenous vegetables of Sudan: production, utilization and conservation. In: Guarino, L. (Editor). Traditional African vegetables. Proceedings of the IPGRI international workshop on genetic resources of traditional vegetables in Africa: conservation and use, 29–31 August 1995, ICRAF, Nairobi, Kenya. Promoting the conservation and use of underutilized and neglected crops 16. pp. 117–121.
- More, T.A., 2002. Enhancement of muskmelon resistance to disease via breeding and transformation. Acta Horticulturae 588: 205–211.
- Oluoch, M.O. & Welbaum, G.E., 1996. Effect of postharvest washing and post-storage priming on viability and vigour of six-year -old muskmelon (Cucumis melo L.) seeds from eight stages of development. Seed Science and Technology 24(2): 195–209.
- Polacchi, W., MacHargue, J.S. & Perloff, B.P., 1982. Food composition tables for use in the Middle East. Food and Agriculture Organization, Rome, Italy. 265 pp.
- Purseglove, J.W., 1968. Tropical Crops. Dicotyledons. Longman, London, United Kingdom. 719 pp.
- Stepansky, A., Kovalski, I. & Perl-Treves, R., 1999. Intraspecific classification of melons (Cucumis melo L.) in view of their phenotypic and molecular variation. Plant Systematics and Evolution 217: 313–333.
- Tindall, H.D., 1983. Vegetables in the tropics. Macmillan Press, London, United Kingdom. 533 pp.
- USDA, 2002. USDA nutrient database for standard reference, release 15. [Internet] U.S. Department of Agriculture, Beltsville Human Nutrition Research Center, Beltsville Md, United States. http://www.nal.usda.gov/fnic/foodcomp. June 2003.
- Wyllie, S.G., Leach, D.N., Wang, Y.-M. & Shewfelt, R.L., 1995. Key aroma compounds in melons: their development and cultivar dependence. In: Rousseff, R.L. & Leahy, M.M. (Editors). Fruit flavors: biogenesis, characterization and authentification. American Chemical Society, Washington D.C., United States. pp. 248–257.
- Zheng, X.Y., Wolff, D.W. & Crosby, K.M., 2002. Genetics of ethylene biosynthesis and restriction fragment length polymorphisms (RFLPs) of ACC oxidase and synthase genes in melon (Cucumis melo L). Theoretical and Applied Genetics 105: 397–403.
Sources of illustration
- Hassib, M., 1938. Cucurbitaceae in Egypt. Faculty of Science, Fouad 1 University, Cairo, Egypt. 173 pp.
- Paje, M.M. & van der Vossen, H.A.M., 1993. Cucumis melo L. In: Siemonsma, J.S. & Kasem Piluek (Editors). Plant Resources of South-East Asia No 8. Vegetables. Pudoc Scientific Publishers, Wageningen, Netherlands. pp. 153–157.
Author(s)
- H.A.M. van der Vossen
Steenuil 18, 1606 CA Venhuizen, Netherlands
- I.M. El Tahir, Plant Genetic Resources Unit, Agricultural Research Corporation, P.O. Box 126, Wad Medani, Sudan
- M.O. Oluoch, AVRDC Regional Center for Africa, P.O. Box 10, Duluti, Arusha, Tanzania
Correct citation of this article
van der Vossen, H.A.M., El Tahir, I.M. & Oluoch, M.O., 2004. Cucumis melo L. [Internet] Record from PROTA4U. Grubben, G.J.H. & Denton, O.A. (Editors). PROTA (Plant Resources of Tropical Africa / Ressources végétales de l’Afrique tropicale), Wageningen, Netherlands.
Accessed 18 December 2024.